Collaboration lies at the heart of the VICT3R project. Bringing together data scientists, software and computer system validation experts, industry partners, and regulators is essential to tackle the complex challenge the project addresses: developing Virtual Control Groups (VCGs) that could one day replace concurrent control groups in animal studies.
On 26–27 February 2026, the VICT3R consortium met in Barcelona for its 4th Consortium Meeting and General Assembly. The meeting was held in a hybrid format, allowing partners to join either in person or remotely. In total, close to 130 participants took part, including over 60 attending in person − a strong sign of the engagement across the consortium and its wider community.
Over two days, partners shared updates, exchanged ideas, and discussed the next steps in the development and implementation of VCGs. As in previous meetings, the atmosphere was lively and interactive, with conversations flowing between disciplines, from toxicology and data science to regulatory science and digital infrastructure.
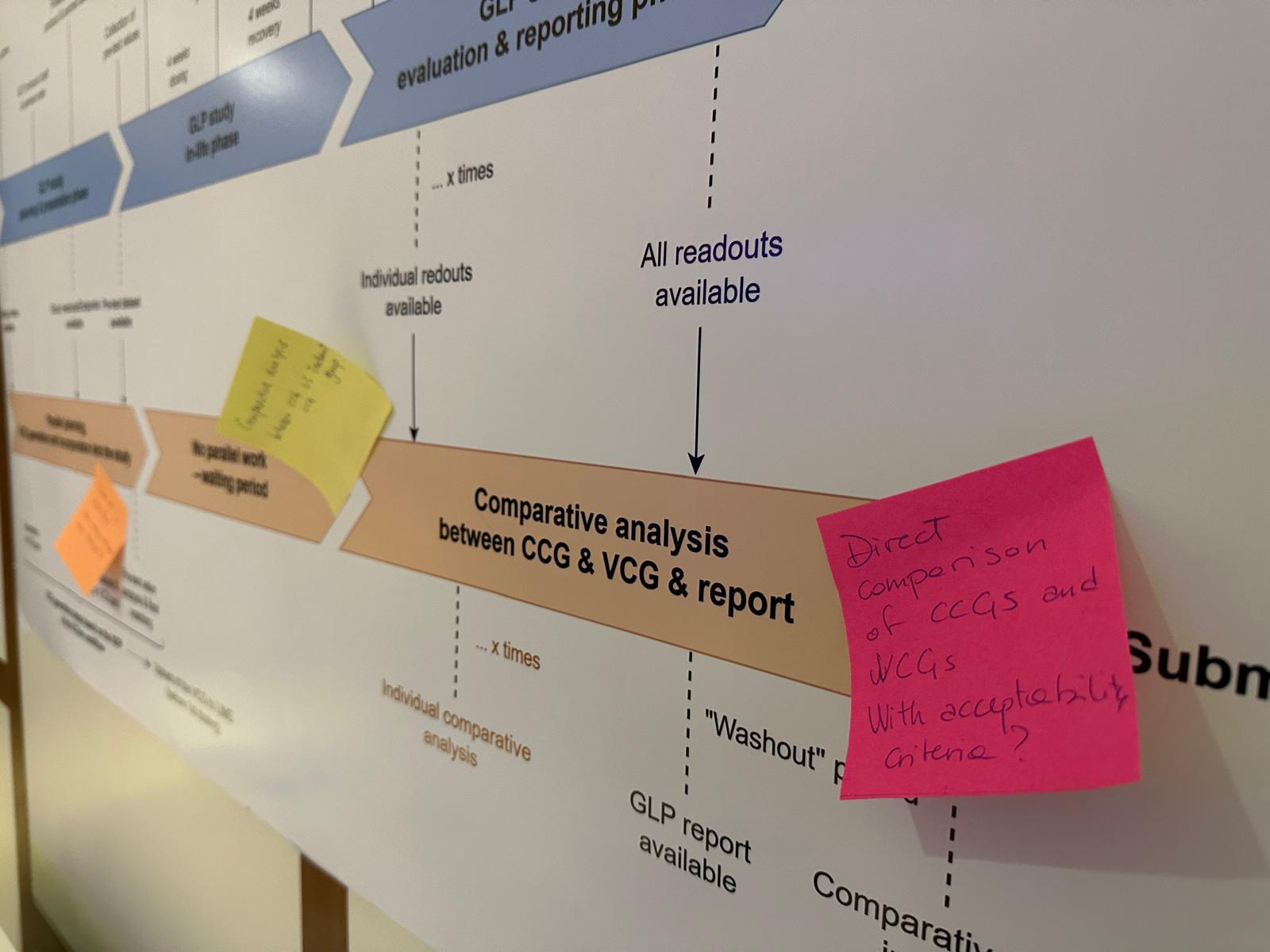
The meeting opened with a satellite session on the planning, conduct, and analysis of prospective and retrospective VCG studies, offering a valuable opportunity for study directors and study monitors to discuss methodological questions and start planning the design and evaluation of upcoming studies including VCGs.
During the plenary sessions, partners highlighted progress across several areas of the project. This included the development of VCGen, a software tool designed to support the generation of VCGs, progress of the VICT3R database alongside advances in industry data contributions, data standardisation, and quality assessment. Discussions also explored how VCG workflows can be integrated into laboratory information management systems, progress in extending the VCG concept to additional study types beyond repeated-dose toxicity, and the integration of whole-slide images into the VCG evaluation.
Regulatory engagement remains a key priority. The consortium reviewed ongoing interactions with EMA and OECD and discussed the next steps needed to support the potential acceptance of VCG approaches in regulatory safety assessments.
Looking ahead, the consortium also discussed future training activities and the identification of potential end-users of VCG tools, recognising that building the necessary expertise will be essential for the successful adoption of these approaches.
Another important discussion focused on data integrity and computer system validation (CSV). Ensuring that digital systems and data processes meet rigorous validation standards is critical for work conducted under Good Laboratory Practice (GLP). Without validated systems and demonstrable data integrity, the regulatory use of VCG-based approaches would not be possible.
The meeting also provided space to reflect on long-term sustainability, exploring how the tools, data resources, and expertise developed within VICT3R can continue to benefit the wider scientific and regulatory communities beyond the project’s lifetime.
The consortium was pleased to welcome Christian Desaintes, Policy Officer from the European Commission, who joined the discussions. The meeting also benefited from the input of the VICT3R Scientific and Regulatory Advisory Board (SRAB). Patience Browne (OECD) and Philip Marx-Stoelting (BfR) participated in person, while other SRAB members joined remotely and shared their perspectives on the project’s progress and future priorities.
As VICT3R moves forward, collaboration across disciplines and sectors will remain essential to translating the concept of Virtual Control Groups into a practical and trusted tool for toxicology research.
A big thank you to everyone in the VICT3R community who contributed to the discussions and helped make the meeting such a productive event. We look forward to welcoming everyone again for the 5th VICT3R Consortium Meeting on 7–8 October 2026, the venue for which will be confirmed in the coming months.